Earth teemed with primitive life more 3½ billion years ago, at a time when the young Sun was too faint to keep our planet from freezing over. New climate models are converging on a solution to this longstanding paradox.
Ah, summer — the season when we're amply reminded that the Sun constantly bathes our planet with 1,367 watts of energy per square meter. And its radiant glow doesn't vary much at all, 0.1% or so throughout an 11-year solar cycle and perhaps as much as 0.5% over many centuries. That's a good thing — life depends on a steady flow of light and heat.
But the Sun was not nearly so shiny in its infancy, when it put out only about 70% of the energy it does today. (Reason: over time, as fusion converts hydrogen to helium, the Sun's core has gotten denser and must burn hotter to counteract collapse due to gravity. The young Sun had less helium, so the core was cooler and the fusion rate less furious.)

Primitive life forms were common on Earth more than 3 billion years ago — a time when the Sun was some 20% fainter than it is today.
Peter Sawyer / Smithsonian Institution
In 1972 Carl Sagan and George Mullen realized that, because of this "faint young Sun," the early Earth should have been completely frozen over — and yet it wasn't, based on all kinds of geologic and paleontologic evidence. In fact, it seems that Earth had liquid oceans and teemed with primitive life by 3½ billion years ago (and likely earlier still).
How Earth managed to avoid becoming the solar system's largest snowball in those Archean times is a climatic paradox that has challenged astrobiologists for decades. The most logical explanation is that our planet's primitive atmosphere contained enough greenhouse gases to boost the surface temperature and keep everything from freezing solid. But which gases? Thanks to the global-warming debate, we all know about the heat-trapping power of carbon dioxide (CO2), which has reached 360 parts per million. But water vapor (H2O) and, especially, methane (CH4) and ammonia (NH3) are greenhouse gases as well.
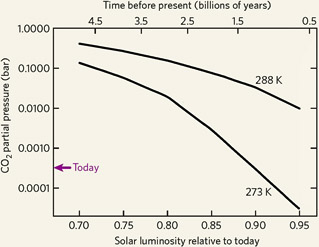
When the Sun formed some 4½ billion years ago, it shone with only about 70% of the intensity it had today. In order for Earth's surface to be at the freezing point of water (273 K) or as warm as it is now (288 K), much more carbon dioxide must have been present in the ancient atmosphere than exists today.
P. von Paris & others
All Earth really needed was an atmosphere pumped up with enough CO2 and H2O. Just how much we don't really know. By one estimate, illustrated at right, 70 times the present level of carbon dioxide would have kept the surface just above freezing (273 K), and it might have required 700 times as much to reach today's global average of 288 K (59°F).
However, geologic evidence is telling a different story, one that caps the CO2 levels at no more than 50 times today's value. In fact, certain banded iron formations imply that the Archean atmosphere might have had no more than about 900 ppm CO2 — just three times the modern-day levels.
So what else, if not abundant CO2, might have been in the greenhouse formula? That's been a complicated and hotly debated issue. Lots of gas mixtures and alternatives have been kicked around over the years. Until his death in 1996, Sagan championed a mix of ammonia and a little methane — gases that might have proved crucial to the development of life. But ultraviolet sunlight breaks down ammonia very rapidly.
Potent methane would have worked in combination with CO2, but too much of it would have led to smoggy hydrocarbon hazes that would have reflected sunlight away, making Earth cooler. As an alternative to touchy gas formulations, in 2010 a research team led by Minik Rosing (Nordic Center for Earth Evolution) proposed that, since early Earth had less landmass, it was darker overall and absorbed more sunlight.
A key problem all along has been the computer-intensive programming needed to simulate long-ago climates. To get their models to run at all, researchers have had to simplify the computations to a single dimension — a vertical profile from Earth's surface to the top of its atmosphere. But these 1D models can't account for circumstances like the role of clouds or the fact that ice forms first at the poles, rather than everywhere at once.
Modern computational horsepower has finally made 3D modeling possible, if only barely. In July's issue of Astrobiology, Eric Wolf and Brian Toon (University of Colorado, Boulder) describe the results of thousands of hours of number-crunching using the university's Janus supercomputer. "Though we use a complex 3D model, our solution is at its heart very simple," Wolf explains. "We only need CO2 and a touch of CH4. No tricks, no high obliquity, no changes to clouds, land, total surface pressure, or exotic greenhouse species."
Wolf and Toon conclude that about 2.8 billion years ago, when sunlight was 20% weaker, an atmosphere with 1.5% CO2 (about 40 times what we have now) and a dash (0.1%) of CH4 would have produced a "hot", virtually ice-free Earth with mean temperatures close to what they are today.
Reducing the carbon dioxide to just 0.5% and eliminating the methane yields a global mean temperature of 260 K (9° F) — well below freezing. But even at this chilly temperature, the 3D modeling suggests that about half of the open ocean would remain ice free. "One key difference with 1D models is that they assume, automatically, that the whole planet must be frozen if the mean surface temperature dips below 273 K," Wolf says. "In 3D we find that this is not the case — large areas of open ocean can persist."
"The faint-young-Sun problem is solved well enough for the time being," observes James Kasting, a Penn State climate specialist who's been wrestling with atmospheric evolution since the 1980s. "But it's one of those problems that will never really be 'solved,' because the parameters for early Earth — particularly the surface temperature — will always be negotiable."
Interestingly, Sagan's ammonia-enriched mix isn't entirely dead. The ammonia might have been protected from destruction by tiny, fractal-shaped particles in a high-altitude organic haze derived from methane. So, if primordial microbes produced lots of methane, conceivably ammonia could have lingered long enough to warm the surface by a few degrees.
Both the ammonia and methane would have disappeared later on, reacting with the oxygen given off by photosynthetic organisms. That led to a temporary drop in temperatures — and episodes of widespread glaciation — but by then the Sun had apparently warmed enough to sustain Earth's emerging lifeline.
 18
18
Comments
tom dasilva
July 18, 2013 at 4:05 pm
what is the time history of earth's gravitational collapse heat dissipation?
You must be logged in to post a comment.
Rod
July 18, 2013 at 6:21 pm
2.8 billion years ago? What about 3.5 billion years ago when Precambrian life was in the rocks and the solar energy even less? The earth's atmosphere must evolve in step with increasing solar energy over these vast spans of time too otherwise we could have another Venus here. Perhaps the fix is on 🙂 Another thought - the earth never orbited a faint young Sun and the geologic record could be telling us that too.
You must be logged in to post a comment.
Tom D
July 18, 2013 at 6:42 pm
Question about the quote "the Sun constantly bathes our planet with 1,367 watts of energy per square meter". Is that per second?
You must be logged in to post a comment.
Tom D
July 18, 2013 at 6:45 pm
I've read before, possibly in Sky & Telescope, that when the Moon first formed it was so close to the Earth that the oceans must have steamed from the tidal energy. Is this true? Could this be part of the 'missing' warmth?
You must be logged in to post a comment.
Rick
July 19, 2013 at 6:20 am
I agree with 2 earlier comments. After gravitational collapse, how quickly did the Earth cool. 3.5 billion years ago there may have been more volcanic activity releasing CO2. Also, Evidence suggests that the moon formed very close to the Earth over 3 billion years ago. It's tidal pull may have been so great to constantly break any ice that tried to form. Models should consider these two variables as well.
You must be logged in to post a comment.
Bruce Mayfield
July 19, 2013 at 7:38 am
Those are some interesting questions Tom D. (1) I Don’t know how fast the earth would have cooled from its initial formation. But since the earth’s outer core is still molten and since volcanism and plate tectonics are still ongoing it’s clear that the interior never has cooled off. This is due to the continuous release of energy from the radioactive decay of elements like Uranium (half life 10 billion years) and its daughter elements. But the further you go back in time, the more radioactive heat would have been produced. Even radioactive Aluminum 26 (hl 717,000 years) was an important heat source in the early solar system. (2) By definition a watt is a unit of power equal to one joule per second or electrically one volt – amp per second. The 1,367 watt per square meter figure must be the total solar flux the upper atmosphere receives. (3) If the moon formed as it is thought via a giant impact about 100,000 years after the earth’s initial formation then it would have started out as a molten mass from the great heat generated by this collision. This collision would have melted or re-melted the earth as well. Liquid water on the warm young Earth would have often been boiled off during volcanic and impact events. And as you mentioned Tom, after the moon’s formation tidal friction would also have been important with the closeness of the early Earth-Moon system. So from the above we can see that the early Earth would have been quite warm in spite of the cool young sun.
You must be logged in to post a comment.
Milton
July 19, 2013 at 10:29 am
Whom ever filled in the bracketed section in this quote:
"Wolf and Toon conclude that about 2.8 billion years ago, when sunlight was 20% weaker, an atmosphere with 1.5% CO2 (about 40 times what we have now) "
should recheck their figures. 1.5% C02 is 15,000 ppm and current atmospheric C02 levels are in the 350 to 400 ppm range (0.035 to 0.04% range). So either the % C02 value of 1.5% (maybe it's 0.15%?) or the 40 time (would be 400) values are wrong.
And the earth was a lot hotter internally back in the Archean times, which is why we have spinefex Komattite ultramafic lava flows in Archean terrain then and none afterwards. The crust must have been very thin, like the scum floating on the top of bubbling pot of soup. A large meteor / asteroid impact would cause the crust to be punctured and massive lava flows would be released.
Possibly are not taking into account the effect of having a moon orbiting so much closer to the earth in the Archean, which would have had a much larger effect on the earth mantle temperatures for internal heating (i.e. as happens with IO near Jupiter). Many of the Archean beach deposits I have seen as a mining geologist in the NWT must have had extremely large tides to create some of the boulder conglomerates (basically bowling ball sized rhyolite rounded clasts floating in carbonate exhalite matrix). Even 1.6 billion years ago, stromatolite colonies were enormous hump mounds up to a few meters in height and 10 meters across at the Carswel lake crater / crypto volcanic event in Northern Saskatchewan, which indicates just how high the tides must have been then.
You must be logged in to post a comment.
Anthony Barreiro
July 19, 2013 at 5:09 pm
This is a fascinating report, and an interesting discussion. There seem to be too many parameters to rule in or out any one possible explanation for the Earth's early warm climate, so everybody gets to entertain their own favorite hypothesis. Toward the end of the article Kelly reports how the oxygen produced by the first plants oxidized organic greenhouse gases, leading to an ice age. So life forms have been influencing the climate for billions of years! Why is it so hard for some people to accept that we humans are causing global warming today?
You must be logged in to post a comment.
Bruce
July 19, 2013 at 5:16 pm
Rod, it seems as though one of your favorite paradoxes might be under serious threat of resolution. In addition to the points already made, consider where much of the Earth’s water evidently came from, the asteroid belt. It has been shown that the isotopic ratios of Earth’s water closely match that of the carbonaceous chondrites (Like the one that exploded over Russia a few months back). To supply all this H2O the Earth must have received a tremendous bombardment over the eons. So to make up for the fainter Sun we have several heat sources at hand, including gravitational contraction, radioactive decay and bombardment by large impactors. Then to hold in the heat we have darker albedo and increased levels of greenhouse gasses. Impactors and volcanic activity both can cause spikes in the amounts of CO2 and H2O vapor, increasing the greenhouse effect. With all this early earthly heating going on, it may in fact be a good thing that the early Sun was fainter, or our planet may have been too hot for life until much later than it appeared. This actually seems like very wise thermal management to me. 🙂
You must be logged in to post a comment.
Bruce
July 19, 2013 at 5:21 pm
And I omitted the very important tidal effects caused by Earth's unusual moon.
You must be logged in to post a comment.
Rod
July 20, 2013 at 6:19 pm
Bruce, I find it interesting all of these comments. Perhaps some should review the baby fat Sun which was larger than our present Sun as the solution - "Baby Fat" on the Young Sun?, http://www.spacedaily.com/reports/Baby_Fat_on_the_Young_Sun_999.html Feb-12. Or perhaps we can look at Earth orbiting closer to the Faint Young Sun - Why Earth Is Not an Ice Ball: Possible Explanation for Faint Young Sun Paradox, http://www.sciencedaily.com/releases/2012/05/120530152034.htm, May-12. Perhaps we could select - “(Phys.org)—Geologists Robin Wordsworth and Raymond Pierrehumbert of the University of Chicago, suggest in a paper published in the journal Science that early Earth was kept warm enough for life to develop by collisions between hydrogen and nitrogen molecules in the atmosphere.", Geologists theorize early Earth was kept warm by hydrogen-nitrogen collisions January 4, 2013 by Bob Yirka, http://phys.org/news/2013-01-geologists-theorize-early-earth-hydrogen-nitrogen.html. Bruce the list goes on. My earlier post here is the answer 🙂
You must be logged in to post a comment.
Edward Schaefer
July 20, 2013 at 8:40 pm
I think that people need to be careful here about such things as extra energy coming from tides, gravitational collapse, and greater internal heating. Yes, all three were greater in the past and much greater in the Earth's early history. But within a billion years, all three would have diminished to relatively low values compared to the Sun's energy flux (even if those values were significantly greater than today's). So you still end up with the faint young Sun paradox at work. I will admit that I lack exact figures, but I do not at all believe that the scientific community would have ignored these inputs all of these years. -- As for this article, I see it as significant progress in resolving this issue. It will be interesting to see what happens as more cases are modeled and other attempt to reproduce these results.
You must be logged in to post a comment.
Bruce
July 21, 2013 at 4:02 am
Rod, your very well referenced comment demonstrates several things. It makes your point that there have been numerous attempts at solving this mystery over the years. And as you say “the list goes on”. I suspect that you may have cherry picked the most outlandish sounding from among them. Your comment indicates that this issue is indeed one that you must be quite fond of following, and your two comments together seem to show a reluctance on your part to even entertain the notion that this problem could even be solvable at all. You state that your earlier post is the answer, wherein you wrote, “the earth never orbited a faint young Sun.” Kelly explained well why the young Sun had to have been fainter in his second paragraph. I don’t think conventional astrophysics is wrong here.
You must be logged in to post a comment.
Bruce
July 21, 2013 at 10:07 am
Edward, you make reasonable points, so I’ll try to slow the pace of my ‘speculative rampage.” But speculations are part of what makes this subject so interesting and could help to insure that all the bases have been covered. And as has been noted this is a very multi-faceted, complex problem. As Anthony pointed out “humans are causing global warming today,” and this is with a much smaller CO2 increase than anything mentioned in this article. This shows that even relatively small changes in our atmosphere can produce significant effects (such as the alarming rate of glacial melt being observed globally). So being able to accurately model all the factors affecting Earth’s temperature budget is quite important. Yes we would hope that all factors have been taken into account, but Kelly informed us that up until this study ‘simplified 1D models’ were being used, which in hindsight sounds rather weak. But since this whole system is so sensitive omitting even a small input could produce suspect results, IMO. I do agree though that the fainter sun would have caused cooler temps in the distant past, or else the periods of massive glaciation, possibly even to the Snowball Earth extent would never have occurred.
You must be logged in to post a comment.
Martin George
July 22, 2013 at 10:17 pm
Regarding the question about wattage, one watt equals one joule per second. So the total amount of energy is in joules, and the RATE of energy is measured in watts.
Martin George
You must be logged in to post a comment.
Tom Yelin
July 28, 2013 at 8:31 am
Kelly writes that the current carbon dioxide concentration is 360 ppm. This is incorrect. It is now 400 ppm (or very nearly that).
You must be logged in to post a comment.
Peter
July 28, 2013 at 5:02 pm
Observations suggest: delta_T = k*log(CO2), where k is a constant. In other words, if doubling CO2 concentration raises Earth's temperature 4 degrees, doubling it again will raise it another 4. A warm world is better than a frozen one. On the other hand, thresholds exist, so we are kind of playing with fire.
You must be logged in to post a comment.
Bruce
July 29, 2013 at 8:47 am
The last two comments from Tom Yelin and Peter are very enlightening, and not only for this thread but also for the one under “The Weakest Solar Cycle in 100 Years” newsblog as well. I hope you don’t mind but I am going to quote your comments into that thread too. I think the points you have made here are important. Thank you for making them.
You must be logged in to post a comment.
You must be logged in to post a comment.