Scientists studying ice here on Earth think they’ve confirmed why comets have hard crusts covered in hydrocarbon gunk. Comets are hodgepodge objects of ice and rock, with relatively “soft” (read: porous) interiors and hard exteriors — so hard, in fact, that the Philae lander from ESA’s Rosetta orbiter broke its hammering tool trying to penetrate the surface of Comet 67P/Churyumov-Gerasimenko. Like several comets before it, Comet C-G’s nucleus has also proven remarkably dark (basically like charcoal) and seems to be coated with organics.
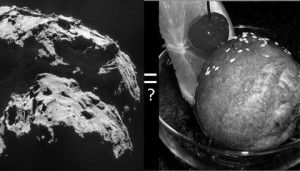
Comet: ESA / Rosetta / NAVCAM; Ice Cream: Diane / Wikimedia Commons
It’s hard to get to a comet, so Antti Lignell (Caltech) and Murthy Gudipati (JPL) explored super-cold ice laced with hydrocarbons in the lab. They watched what happened as ice transitioned from an amorphous state — in which ice crystals are stuck together haphazardly — to a harder, crystalline state as they heated it. (Only crystalline ice, with its ordered lattices, exists naturally in Earth conditions.) The scientists report in Journal of Physical Chemistry A that, as the ice went from amorphous to crystalline, it coughed up its hydrocarbons as aggregates, making space for the ice to build its stiff lattice.
This process is kind of like how brine freezes: the ice doesn’t incorporate salt molecules into its lattice, so the residual liquid just gets saltier and saltier.
These results support what many researchers assume: there are deep deposits of fluffy organics on periodic comets’ surfaces, explaining their dark coloring. And the surface that Philae tried to drill into was certainly harder than amorphous water ice. But even if in approaching the Sun the comet builds up a crystalline ice crust and hydrocarbon surface layer, it’s still unclear what happens when things get too hot for the ice altogether: comets may lose a meter in depth as sunlight sublimates the ice, so whether the comet would shed this crusty stuff and what that would do to the situation isn’t known.
You can read more about the result in JPL’s press release, “Why Comets Are Like Deep Fried Ice Cream.” (No really, the metaphor works — although I think chocolate would taste a whole lot better than the gunk on 67P.)
Reference: A. Lignell and M. Gudipati. “Mixing of the Immiscible: Hydrocarbons in Water-Ice near the Ice Crystallization Temperature.” Journal of Physical Chemistry A. October 10, 2014.
 3
3
Comments
Peter Wilson
February 16, 2015 at 1:26 pm
In technical terms, they're thick-skinned.
You must be logged in to post a comment.
February 16, 2015 at 9:45 pm
As popular at the dirty snow ball concept is, I am sorry, the data supports frosty asteroids.
You must be logged in to post a comment.
February 22, 2015 at 3:21 pm
Comets are hard rocks or iron that carry a positive electrica charge. Because the spacecraft made electrical contact with the positive comet core, the batteries drained and the European comet probe failed. I told Fred Whipple at least 10 years ago that comets were not dirty snowballs. He replied by saying, "You give me an upset stomach." Now we have wasted an 11 year mission on an incorrect comet model. I am sorry for Dr. Whipple!
You must be logged in to post a comment.
You must be logged in to post a comment.