New research suggests that an exoplanet's atmosphere can contain lots of oxygen — none of it created by life — if bathed in the wrong mix of ultraviolet radiation from its host star.
Fact: free oxygen shouldn't exist in Earth's atmosphere. It's orders of magnitude out of equilibrium with the other gases in the mix. But, thanks to the rise of photosynthetic organisms, its abundance started increasing some 2.2 to 2.4 billion years ago, and (thankfully) it's here to stay.

An artist's concept of planets orbiting the cool, red dwarf star Gliese 876, located 15 light-years away in the autumn constellation Aquarius.
NASA / Greg Bacon (STScI)
This same scenario is what many astrobiologists hope has happened on Earth-like worlds circling other stars, particularly M dwarfs. These stars are preferred for exoplanet searches because they're rampantly abundant in our galaxy, have very long stable lifetimes, and have the cool, dim, and low-mass characteristics that make finding smallish planets around them relatively easy.
Cosmic oddsmakers recently put the chance of finding an Earth-like planet around an M dwarf at nearly 50%. And the presence of oxygen in its atmosphere would be taken as a strong indication of abundant biological activity.
Well, maybe not, say Feng Tian (National Astronomical Observatories of China) and four colleagues from the U.S. and Argentina. At today's meeting of the American Astronomical Society's Division for Planetary Sciences, Tian showed how planets circling in the habitable zones of M dwarf could have lots of free oxygen but be completely lifeless.
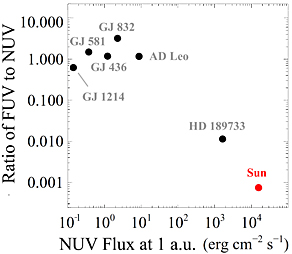
A sample of six dwarf stars studied in 2011-12 with the Hubble Space Telescope shows that they emit orders of magnitude less near-ultraviolet (NUV) radiation than the Sun does — an important characteristic for driving photochemistry of any planets around them.
K. France & others / Astrophysical Journal
The key, he explains, is what's coming off the host star. Earlier this year, a group led by Kevin France (University of Colorado) found that six M dwarfs studied with the Hubble Space Telescope all produce lots of ultraviolet radiation in the regions above their their otherwise subdued photospheres. They crank out a Sun's worth of potent far-ultraviolet energy — but no more than 0.1% of the Sun's near-ultraviolet rays.
That strongly skewed ratio, says Tian, makes all the difference in cooking up compounds an exoplanet's atmosphere. Specifically, the far-UV photons can break up carbon dioxide (CO2) to yield oxygen atoms and, eventually, oxygen molecules (O2) and ozone (O3). Other byproducts from reactions with hydrogen include H2O2 and HO2 (note: not H2O). But there are far too few near-UV photons to keep the reactions going in a way that would reconstitute the CO2. So ultimately the oxygen levels build up.
Oxygen is relatively easy to detect spectroscopically, and it's easy to envision future spacecraft capable of detecting it in exoplanet atmospheres. So will oxygen's presence always be a "false positive" for biologic activity? No, but as Tian cautions, "We need to know more about the ultraviolet environment before claiming the existence of life on exoplanets."
 6
6
Comments
Peter W
October 7, 2013 at 5:23 pm
Why…are they emitting so much more FUV than NUV?
You must be logged in to post a comment.
jason ngim
October 8, 2013 at 8:46 am
Our deep sea volcanoes which are spewing sulphur n other gasses are feeding n sustaining life forms like shrimps n mussels. and they are not relying on oxygen to breath. so yeah, oxygen should not be the key to find life, but the key to sustain us living in that environment, maybe.
You must be logged in to post a comment.
Richard
October 9, 2013 at 7:32 am
To Jason Ngim:
Even deep sea shrimp and mussels utilize oxygen in their respiration. Both use gills to take dissolved oxygen from the water, just like fish. If it's in the Kingdom Animalia or Plantae, it utilizes oxygen for respiration.
There are anaerobic bacteria that don't need oxygen to survive, but your comment is inaccurate. Even plants utilize oxygen for respiration at night when photosynthesis isn't occurring.
You must be logged in to post a comment.
Quizzical
October 9, 2013 at 6:11 pm
Actually, no one should "claim the existence of life on any planet before there is a specimen to study. Too many folks seem to have a hard time differentiating between real science and imaginary notions. They let their imagination run wild and the result is pure nonsense. Captain Kirk will always remain a fictional character and "cruising the galaxy" will have to wait until after we devise a perpetual motion machine. Oh, but wait. There is just such a thing developed in India where a light weight car is reported to run on compressed air and the claim is that soon they will be driving a compressor with compressed air to keep the cycle going. Isn't that wonderful? I know some folks who get all excited about stuff like that but are bored with real science. That says a lot about their intelligence and the quality of their education!
You must be logged in to post a comment.
NS
October 11, 2013 at 6:17 pm
Even if it exists, we won't be able to get a specimen of alien life for decades (if it's elsewhere in the solar system) or most likely centuries if it's in other stellar systems. Until then we'll have to rely on remote observations and general astronomical/biological theory to form any estimate at all on whether life is present on distant worlds. That doesn't make it unscientific or in the same realm as perpetual motion machines. Thinking that it does shows a very limited understanding of science.
You must be logged in to post a comment.
mark
October 17, 2013 at 2:31 am
Nobody's going to anounce a life detection based on only a free oxygen measurement. If there are sufficiently numerous complimentary markers such as abundant methane, spectrum of chlorophyll, CFCs and all the other markers that would be difficult to explain in the absence of life (at whatever level) then that would combine to push for life being there.
This article points to new knowledge that will help us in future detections rather than hinder us by ruling out the possibility of ever being able to determine if ExSo planets harbour life beneath any oxygen rich atmospheres.
You must be logged in to post a comment.
You must be logged in to post a comment.